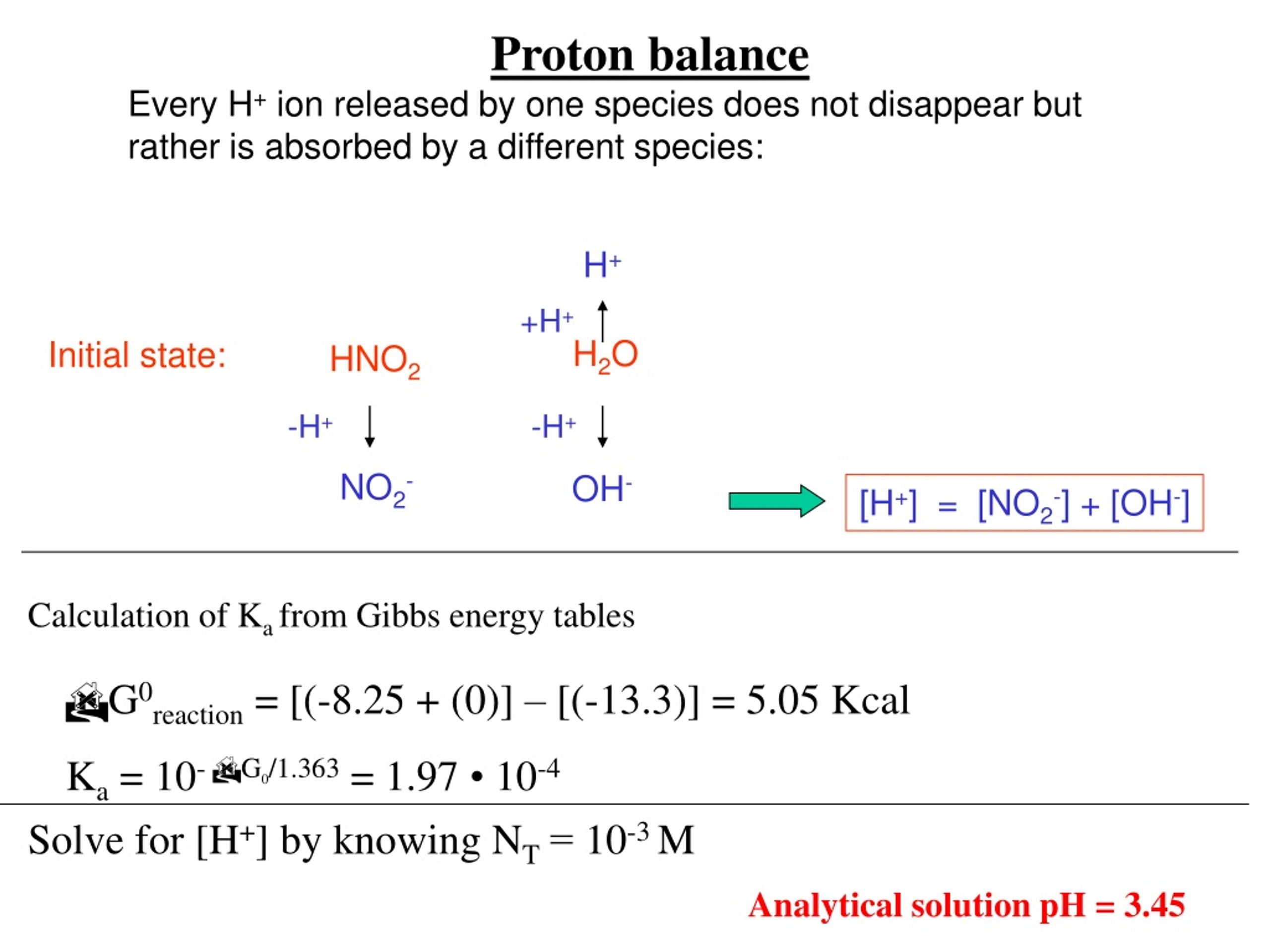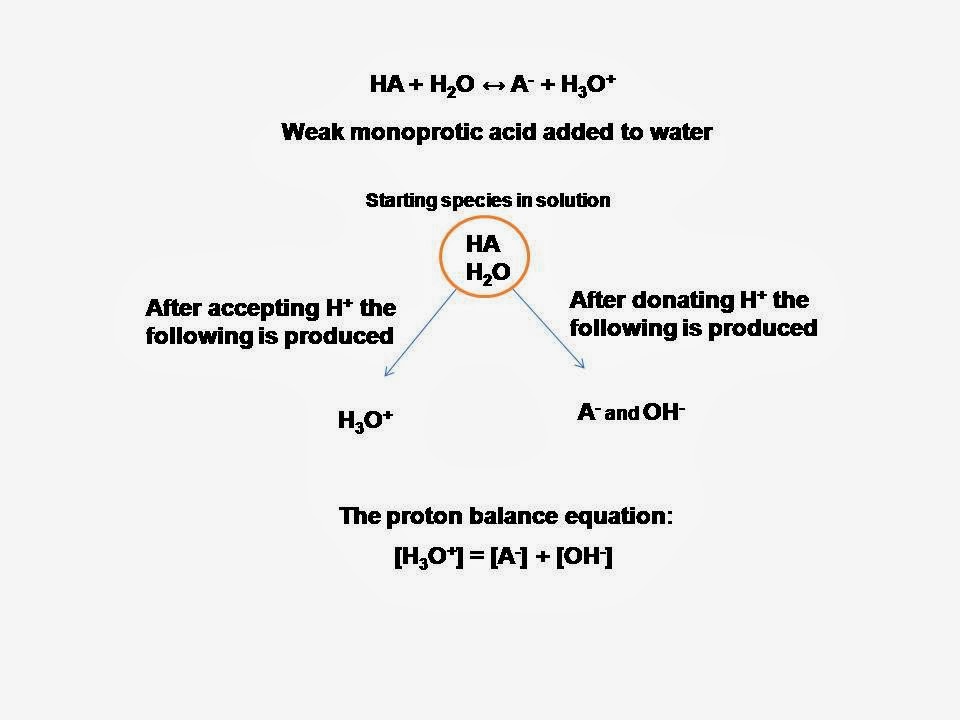
pH calculations and more in fundamentals of pharmaceutics. : Proton balance equation for weak monoprotic acids.

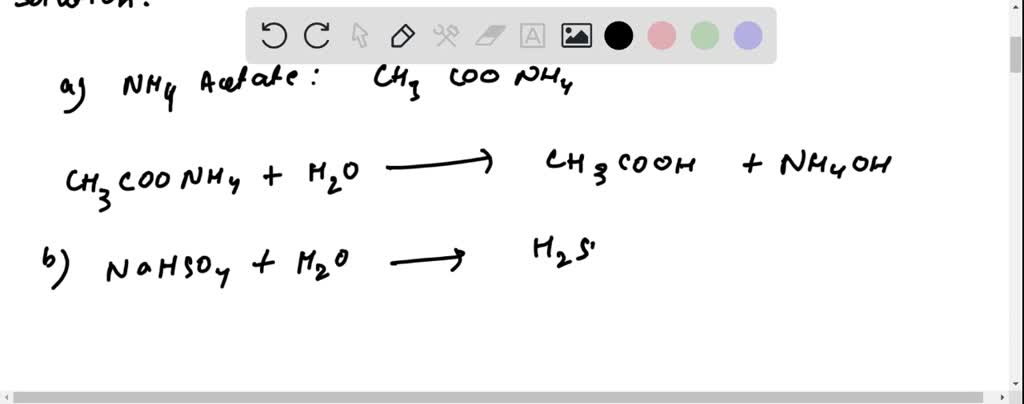
SOLVED: 1. Write down the mass balance, charge balance and proton balance equations for the following solutions: a. NaHC2O4 b. K3PO4 c. NH4HCO3 d. 0.1 M NaHCO3 e. 0.1 M H2CO3

Proton balance of the various types of N fertilizers: a plus indicates... | Download Scientific Diagram

Tautomeric Equilibrium Revisited: proton-tautomerism in solvent and the fundamentals of molecular stability prediction. | Biological and Medicinal Chemistry | ChemRxiv | Cambridge Open Engage